Each amino acid has both a one-letter and three-letter abbreviation. The structures of the 20 amino acids commonly found in proteins are shown in Figure 1. These side chains confer different chemical, physical, and structural properties to the final peptide or protein. 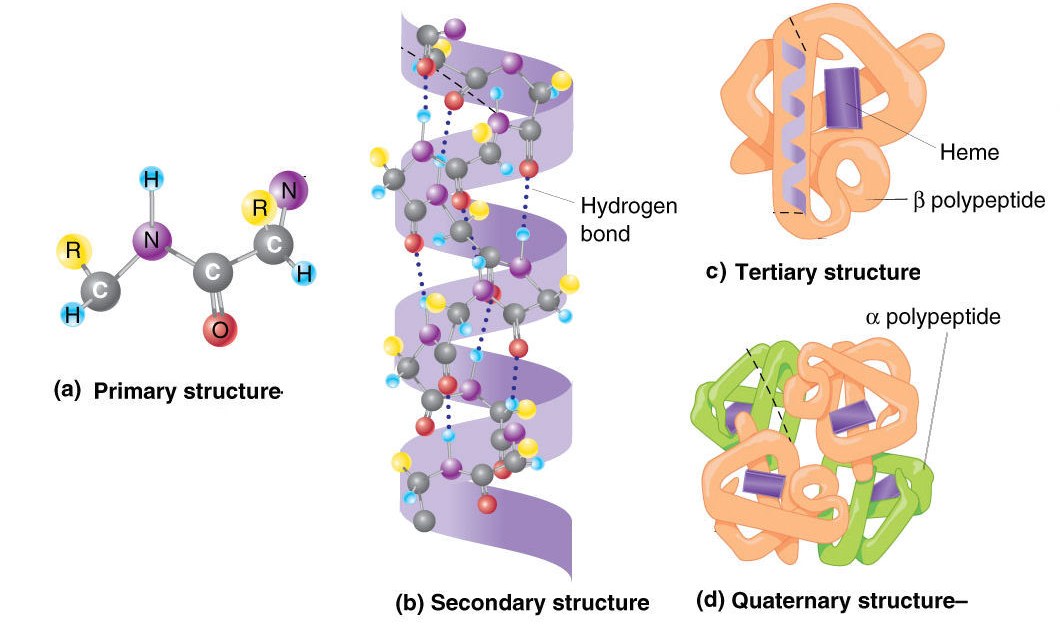
The amino acids differ in structure by the substituent on their side chains. The terms, amino-terminus and N-terminus, describe the end of the sequence with a free α-amino group. The end of the peptide or protein sequence with a free carboxyl group is called the carboxy-terminus or C-terminus. A protein can be made up of one or more polypeptide molecules. Sequences with fewer than 50 amino acids are generally referred to as peptides, while the terms, protein and polypeptide, are used for longer sequences. 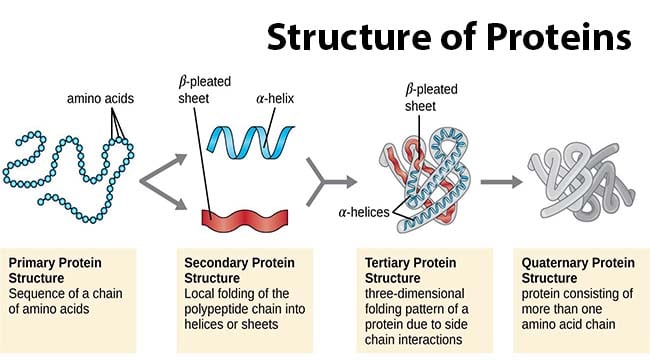
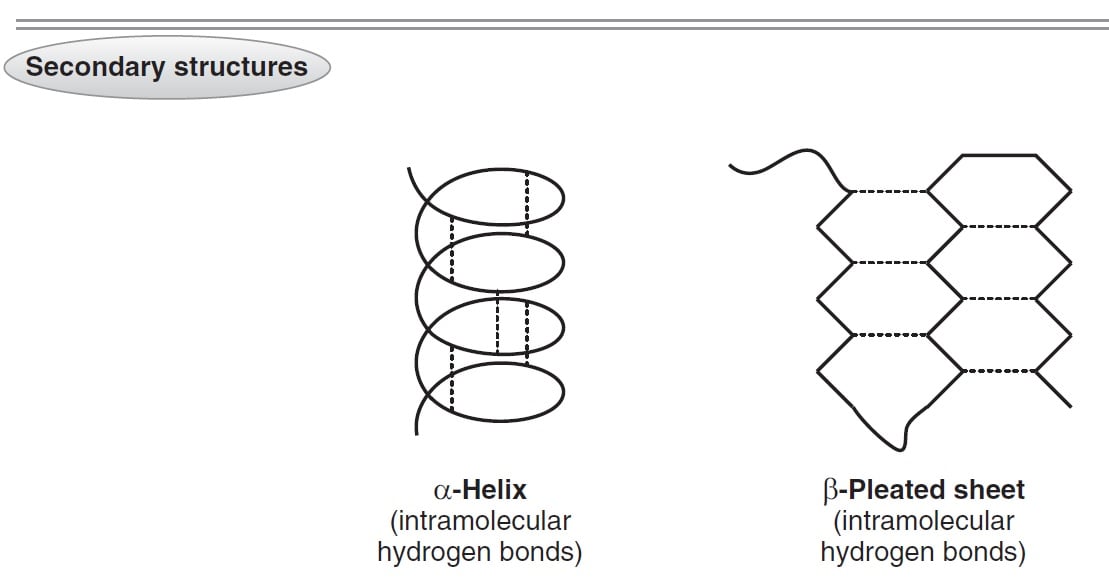
This difunctionality allows the individual amino acids to join in long chains by forming peptide bonds : amide bonds between the -NH 2 of one amino acid and the -COOH of another. Amino acids, as their name indicates, contain both a basic amino group and an acidic carboxyl group. There are 20 different standard L-α-amino acids used by cells for protein construction. Proteins are macromolecules and have four different levels of structure – primary, secondary, tertiary and quaternary. The term, structure, when used in relation to proteins, takes on a much more complex meaning than it does for small molecules. It will also cover briefly how protein structure can be affected during formulation and some of the analytical methods which can be used both to determine the structure and analyze the stability of the protein. This technical brief aims to give the reader a quick overview of protein structure. Formulation of a protein drug product can be quite a challenge, and without a good understanding of the nature of protein structure and the conformational characteristics of the specific protein being formulated, the results can be ruinous. Increasingly, drug developers are looking to large molecules, particularly proteins, as a therapeutic option. Implantable Drug Delivery Systems and Combination Products Dosage Forms.Solubility & Bioavailability Enhancement.An important goal of molecular medicine is to identify biomarkers such as proteins and/or modifications to proteins whose presence, absence, or deficiency is associated with specific physiologic states or diseases ( Figure 4–1). A typical protein is “born” at translation (see Chapter 37), matures through posttranslational processing events such as selective proteolysis (see Chapters 9 and 37), alternates between working and resting states through the intervention of regulatory factors (see Chapter 9), ages through oxidation, deamidation, etc (see Chapter 58), and “dies” when degraded to its component amino acids (see Chapter 29). Proteins are subject to physical and functional changes that mirror the life cycle of the organisms in which they reside. Receptors enable cells to sense and respond to hormones and other extracellular cues (see Chapters 41 and 42). Enzymes catalyze reactions that generate energy, synthesize and degrade biomolecules, replicate and transcribe genes, process mRNAs, etc (see Chapter 7). Hemoglobin transports oxygen (see Chapter 6), while circulating antibodies defend against foreign invaders (see Chapter 52). Actin and myosin filaments form the contractile machinery of muscle (see Chapter 51). An internal protein network, the cytoskeleton (see Chapter 51) maintains a cell’s shape and physical integrity. Proteins are physically and functionally complex macromolecules that perform multiple critically important roles. Outline three strategies for resolving individual proteins and peptides from complex biologic samples to facilitate their identification by MS.Ĭomment on the contributions of genomics, computer algorithms, and databases to the identification of the open reading frames (ORFs) that encode a given protein. Name four chromatographic methods commonly employed for the isolation of proteins from biologic materials.ĭescribe how electrophoresis in polyacrylamide gels can be used to determine the purity, subunit composition, relative mass, and isoelectric point of a protein.ĭescribe the basis on which quadrupole and time-of-flight (TOF) spectrometers determine molecular mass.Ĭompare the respective strengths and weaknesses of DNA cloning and mass spectrometry (MS) as tools for determining protein primary structure.Įxplain what is meant by “the proteome” and cite examples of its potential significance.ĭescribe the advantages and limitations of gene chips as a tool for monitoring protein expression. 
After studying this chapter, you should be able to:Ĭite three examples of posttranslational modifications that commonly occur during the maturation of a newly synthesized polypeptide. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
